Pedro Castillero, Victor Rico-Gavira, Carmen López-Santos, Angel Barranco, Virginia Pérez-Dieste, Carlos Escudero, Juan P. Espinós, and Agustín R. González-Elipe
The Journal of Physical Chemistry C, 121 (2017) 15719−15727
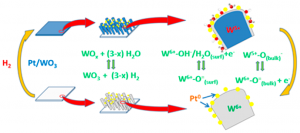
M/WO3 (M = Pt, Pd) systems formed by a porous WO3 thin film decorated by metal nanoparticles are known for their reversible coloring upon exposure to H2 at room temperature. In this work, this gasochromic behavior is investigated in situ by means of near-ambient photoemission (NAPP). Pt/WO3 systems formed by very small Pt nanoparticles
(10 ± 1 nm average size) incorporated in the pores of nanocolumnar WO3 thin films prepared by magnetron sputtering at an oblique angle have been exposed to a small pressure of hydrogen at ambient temperature. The recorded UV−vis transmission spectra showed the reversible appearance of a very intense absorption band responsible for the blue coloration of these gasochromic films. In an equivalent experiment carried out in the NAPP spectrometer, W 4f, O 1s, Pt 4f, and valence band photoemission spectra have been recorded at various photon energies to follow the evolution of the reduced tungsten species and hydroxyl groups formed upon film exposure to hydrogen. The obtained results are compared with those of a conventional X-ray photoemission study after hydrogen exposure between 298 and 573 K. As investigated by NAPP, the gasochromic behavior at 298 K is accounted for by a reaction scheme in which hydrogen atoms resulting from the dissociation of H2 onto the Pt nanoparticles are spilt over to the WO3 substrate where they form surface OH−/H2O species and subsurface W5+ cations preferentially located in buried layers of the oxide network.

